I love the way you overexcite
Or, inflammation and mental health, again.

But first… Yo, a new arch dropped!

Should’ve screenshotted myself, we’re on step 6 now…
Using STAR genome - hierarchical NN architecture encoding
Evolutionary algorigthm to improve the network architecture for a particular case
Cache ⬇️90% compared to transformers and ⬇️37% compared to hybrid models (e.g. Mamba-based)
Parameter count: ⬇️13%
(infer slowly slowly slowly getting faster once I start inferring it’s really hard to stop)
Anyway, putting the bets on Liquid AI as both their liquid network and STAR are good (IMO):

So the mom and pa were right?
Exclusion of any food group of [meat, fish, vegetables] ➡️ increase of depressive symptoms Odds Ratio, adjusted for age, sex, sociodemographics etc.:
🚫 Meat → 1.37 [1.24–1.52]
🚫 Fish → 1.40 [1.31–1.50]
🚫 Vegetables → 1.71 [1.49–1.97]
🍣 Pesco-vegetarian → 1.43 [1.19–1.72]
🍳 Lacto-ovo-vegetarian → 1.36 [1.09–1.70]
Full table:

Don’t deprive yourself of more than 1 product:

- A reasonable Reddit comment: where’s ketogenic in here?

That, and kynurenine effect (wrote on it while postCOVID bitchslapped everyone):
⬆️ Kynurenic acid (⬇️ excitability + neuroprotective + decreased in depression and bipolar + antagonist at NMDA, AMPA, Kainate receptors)
⬇️ Kynurenine (decreased in bipolar + increased in depression, schizophrenia and Alzheimer’s + metabolizes into potentially excitotoxic Quinolinic acid)

And, huh (don’t forget the 6%/year kidney stone risk, and time accordingly)

So, bipolar…
Lithium is modulatory. How exactly?

Been rewriting a lithium overview before, time to add some more insights.
Mechanisms of lithium, revisited
Inhibits GSK-3b (influences metabolism and the whole nervous system cell lifecycle) → neuroprotection and normotimic effects. But you’re not here for that, right?
⬆️ glutamate reuptake with chronic use
⬇️ postsynaptic NMDA activation ← ⬇️ phosphorylation (papa ATP gives the P to tha cell, and she wan the P so bad ayy)

⬇️ excess dopamine signalling in PFC (presumably akin to what aripiprazole does?)
Inflammation/autoimmune and mental health
A recent study had shown 48.1% of bipolar patients have autoimmune or allergic diseases
Cytokines influence kynurenine pathway at large in a complex interplay:

Cytokines can also influence BBB permeability → antibodies and molecular mimicry agents going in and out like it’s their home
Different studies incl. Modabbernia et al. (2013) found that bipolar has:
⬆️ IL 2,4,6, TNF-α, sIL-6R
Mania → ⬆️ IL-1 antagonist (IL-1RA), sIL-2R & sTNFR1
Depression → ⬆️ IL-10 (weird as it’s immunomodulatory)
⬆️ IL-1β & kynurenic acid in spinal fluid
Post-mortem: ⬆️MyD88, NF-κB → IL-1 pathway activators
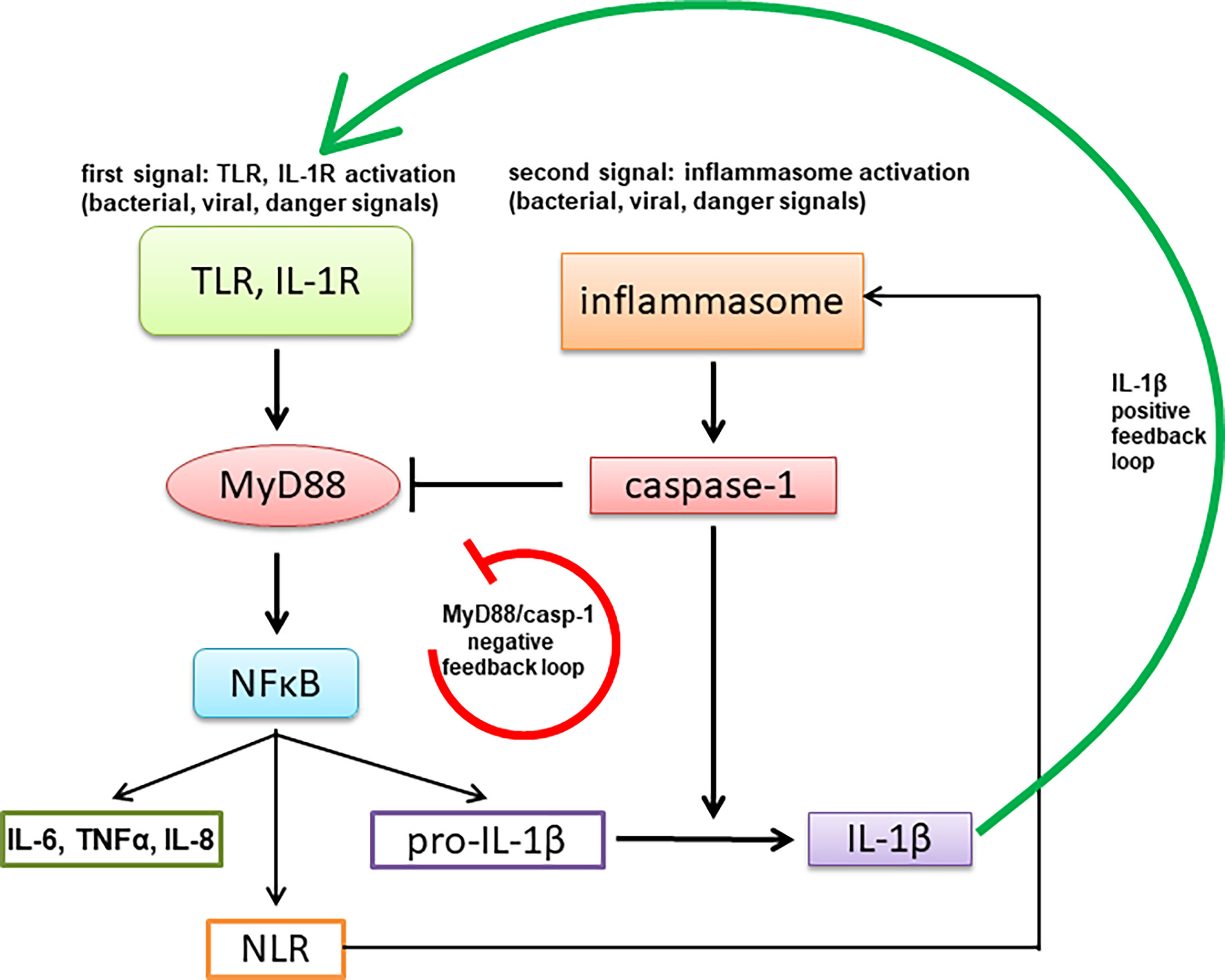
⬆️ PDE4B → ⬆️ inflammation due to cAMP degradation
⬆️ neutrophil/lymphocyte ratio, ⬆️ RBC/lymphocytes during mania
Lithium and immunity (likely downstream effects)
⬆️ IGF-1 expression in lithium responders. IGF-1 inhibits apoptosis → ⬆️ cell survival
⬆️ TNF-α & IL-4 after 8 weeks of lithium
⬆️ IL-1β and ⬇️ IL-6 after 6 months of lithium
Conclusion (criminally simplified)
⬆️ Inflammation → ⬆️ Involvement of kynurenine pathway → ⬆️ Quinolinic acid ⬇️ 5-HTP ⬇️ Kynurenine → (chance of) ⬆️ Excitability
PVT calcium intake/myto dysfunction and bipolar
- Tadafumi Kato, a neuroscientist at Juntendo University in Tokyo
“In mice, the neurons in the PVT branch off into both the amygdala, influencing negative emotions like fear, and to the nucleus accumbens, influencing positive emotions linked to reward,” says Kato.

“If so, it would make sense that overexcitement of the PVT would play a central role in the extreme mood swings associated with bipolar disorder,” he says.
- Mie Kubota-Sakashita
⬆️ blood cell calcium in bipolar
🥴 altered mitochondrial calcium uptake in bipolar
Conclusion (far-fetched)
Dysfunctional mitochondrial function in PVT (Paraventricular nucleus of the thalamus) → ⬆️ calcium uptake → bipolar
Limit overexcitability and…?
As I’ve mentioned before, overexcitability brings a lot to the table - and almost never that’s tasty stuff, mostly nasty.
Things start to look that overexcitability / inflammation / mitochondrial dysfunction in separate brain regions (especially PVT given its connectivity) can lead to a lot of trouble: bipolar, schizophrenia, ASD, ADHD etc.
So, I’d say we can expect four drug MoA patterns emerging:
directly limiting excitability or increasing plasticity → acting on ion channels or glutamate/glycine/ampakine/kainate systems, e.g. modulators like rapastinel/apimostinel/zelquistinel. Just have a look at, say, list of investigational antidepressants
mitochondrial enhancers → overmitochondriating the possible deficits
targeted gene therapy once potential targets are sifted out
anti-inflammatory or anti-kynurenine-pathway-craps treatment
Memantine and bipolar...

LAMotrigine + MEMantine → ⬆️ antidepressive effects and faster onset compared to LAM + PLAcebo
This bad boy can potentially ⬆️ kynurenic acid (the good one) production
And ADHD…
Disrupted melatonin and ADHD

⬆️ IL-6 in ADHD
Melatonin → ⬇️ IL-6 → any defects in melatonin secretion would lead to an increase in inflammation among other things
Welcome to Teleogenic❣️
Other places I cross-post (not always) to: