I've got the power!
Nuclear, since the 1910s...

What the fuss fuse?
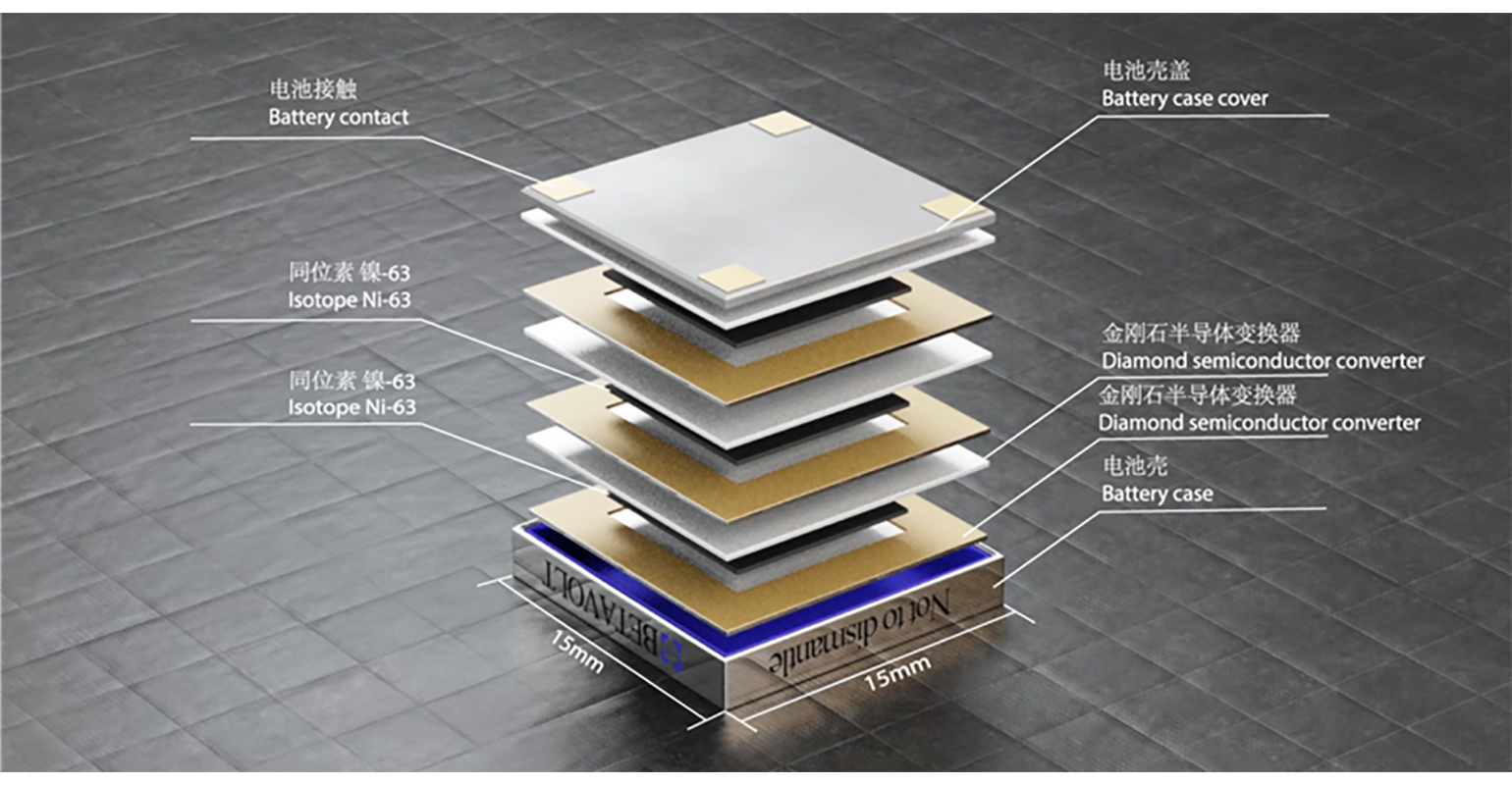
So, you’d likely heard about Betavolt - a revolutionary, game-changing technology: layered battery with a Ni-63 isotope that emits power for up to 50 years! Some facts:
🔋 It should really work for ~50 years as Ni-63’s half-life is 100.1 years
☢️ Radiation-wise it’s rather safe, i.e. it’s beta-radiation + don’t try to open it or make a smoothie. Making a smoothie out of a Li-Pol battery will not do your health much good, too.
🦿 Converts to copper-63 (stable) over time, so it’s extremely environmentally friendly in the long run

👷 Works like this (simple explanation so you’ll be able to engage in broscience wars below):
Betavoltaic batteries comprise two parts: a radioactive emitter and semiconductor absorber. As the emitter naturally decays, high-speed electrons (aka beta particles) strike the absorber. This creates an “electron-hole” pair, which generates a small-but-stable supply of usable electric current.
In simpler words and likely with a lot of mistakes:
electron-hole pair is a complex of an electron (negative charge) and a ‘vacant’ place where an electron should be (positive charge)
when it’s generated near a PN junction, electron is pulled towards N, and the ‘hole’ is pulled towards P
the resulting separation causes the charge to flow (electrons moving)

What’s the game?
“So game-changing”, “Much power”, “Eternal memes” - the media lauds Betavolt. But are they first?
NDB (Nuclear Diamond Battery) is already in the market, claiming 28,000 years of battery life
Radioactive decay-based batteries had been designed since 1913, by Henry Moseley (he’s a one smart chap, innit?)
There were numerous betavoltaic battery startups and techs (incl. using other isotopes like tritium), but they’re in stealth or death mode (we don’t hear much)
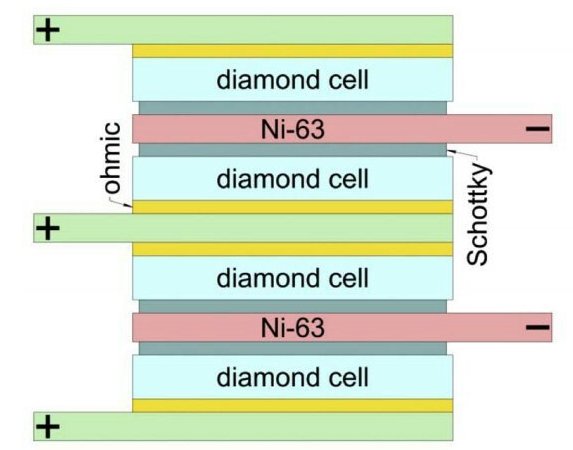
Researchers from MIPT (Moscow Institute for Physics and Technology) had, erm, achieved ~this structure in 2018 (nickel-63 layered with diamond semiconductors)
I’ve honestly tried to find differences between their tech, but couldn’t even with the help of digital hallucinating monsters!
Isotope: Both use nickel-63 as the radioactive source
Semiconductor: Both use diamond as the semiconductor material12
Thickness: Both identify 2 micrometers as the optimal thickness for nickel-63
What are the implications?
As the tech is not new, but it’s definitely a huge market and strategic advantage, we could infer something like:
Conversion efficiency is a huge factor - it’s the main thing changing in this tech AFAIS
Compatibility with current standards - e.g. an AA/CR2032 battery that never has to be recharged is good, but a working safe pacemaker, or a sensor, or a drone - that’s on a whole another level
Sustainability - e.g. are the parts easily sourced/produced/recycled? Are the reagents easy to get or synthesize?
Outreach and adoption - i.e. the ability to secure contracts, synergies (medtech, military, surveillance, corporate etc.)
It looks like China may have the upper hand in government-level large-scale adoption of emerging tech - and the skill of announcing loudly, as can be seen by DeepSeek’s, Unitree’s and now - Betavolt’s releases.
The resulting implications? I wouldn’t want to see a trade/strategic war, but the scientific cold one is both good (more scientific progress - likely better lives on average) and bad (snatching enablers, e.g. Taiwan/TSMC…)

Welcome to Teleogenic❣️
Other places I cross-post (not always) to: